|
five pairs of D/L enantiomers called allose, altrose, glucose, mannose, gulose, idose, galactose, and talose.īelow are the Fischer projects 3-6 carbon aldoses. 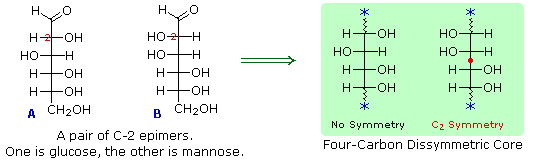
Aldohexoses have sixteen (2 4) possible stereoisomers. 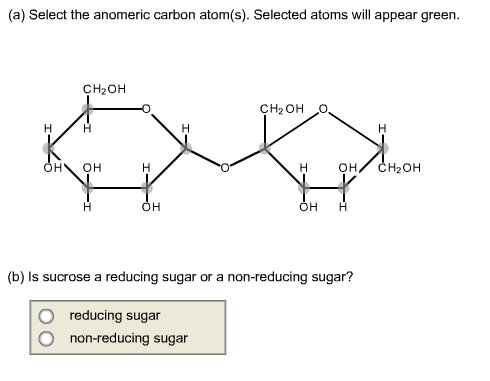
Four pairs of D/L enantiomers called ribose, arabinose, xylose, and lyxose.Īldohexoses: 6 carbon sugars with four chiral centers. Aldopentoses have eight (2 3) possible stereoisomers. Two pairs of D/L enantiomers called erythrose and threose.Īldopentoses: 5 carbon sugars with three chiral centers. 
Aldotetroses have four (2 2) possible stereoisomers. A pair of enantiomers called D-gylceraldehyde and L-glyceraldehyde.Īldotetroses: 4 carbon sugars with two chiral centers. Aldotrioses have two (2 1) possible stereoisomers. To illustrate using present day knowledge, Fischer projection formulas and names for the D-aldose family (three to six-carbon atoms) are shown below, with the asymmetric carbon atoms (chiral centers) colored red.Īldotrioses: 3 carbon sugars with one chiral center. The mirror images of these configurations were then designated the L-family of aldoses. Consequently, Fischer made an arbitrary choice for (D)-glucose and established a network of related aldose configurations that he called the D-family. At the time Fischer undertook the glucose project it was not possible to establish the absolute configuration of an enantiomer. His successful negotiation of the stereochemical maze presented by the aldohexoses was a logical tour de force, and it is fitting that he received the 1902 Nobel Prize for chemistry for this accomplishment. This challenge was accepted and met in 1891 by the German chemist Emil Fischer. These would exist as eight diastereomeric pairs of enantiomers, and the initial challenge was to determine which of the eight corresponded to glucose. The four chiral centers in glucose indicate there may be as many as sixteen (2 4) stereoisomers having this constitution. draw the Fischer projection of D-glyceraldehyde, D-ribose and D-glucose from memory.The epimeric carbon in anomers are known as anomeric carbon or anomeric center. draw the structures of all possible aldotetroses, aldopentoses, and aldohexoses, without necessarily being able to assign names to the individual compounds. C-1 if they are aldoses or in the configuration at C-2 if they are ketoses.When the pairs are enantiomers the prefix becomes ent-.\)Īfter completing this section, you should be able to In chemical nomenclature one of the epimeric pairs is given the prefix epi- for example in quinine and epi-quinine. 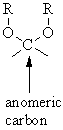
These two molecules are epimers but not anomers. The hydroxyl group in β-D-glucopyranose is equatorial (in the "plane" of the ring) while in β-D-mannopyranose the 2 hydroxyl group is axial (up from the "plane" of the ring). In this case β-D-glucopyranose and β-D-mannopyranose are epimers because the differ only in the stereochemistry at the 2 position. These two molecules are both epimers and anomers. In β-glucose, the -OH group is oriented in the same direction as the methylene group. In α-glucose, the -OH group on the first (anomeric) carbon is in the direction opposite the methylene group. Stereoisomers include enantiomers and diastereomers, both which contain a stereogenic center (excluding geometric isomers, which is a class of diastereomers).įor example, the sugars α-glucose and β-glucose are epimers. In chemistry, an epimer is a stereoisomer of another compound that has a different configuration at only one of several stereogenic centers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
